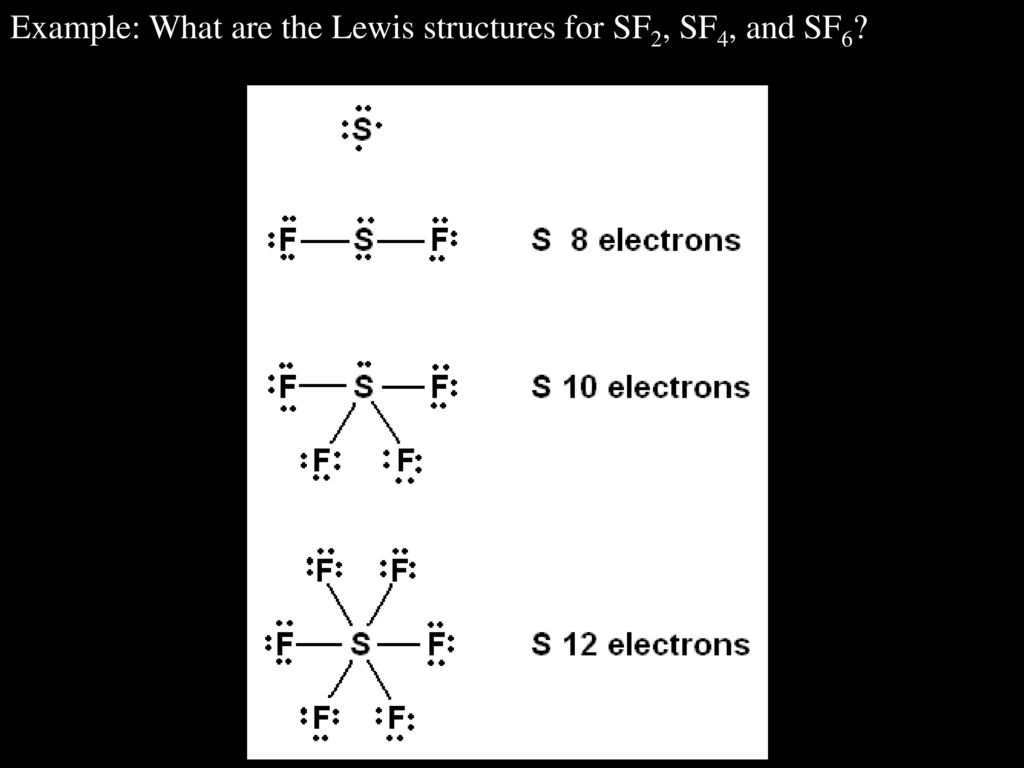 
A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons:įigure 1 shows the Lewis symbols for the elements of the third period of the periodic table. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. In this section, we will explore the typical method for depicting valence shell electrons and chemical bonds, namely Lewis symbols and Lewis structures. In all cases, these bonds involve the sharing or transfer of valence shell electrons between atoms. 
Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
